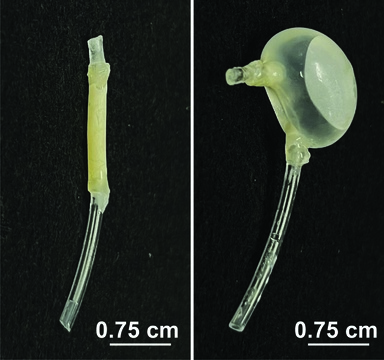
Innovative approaches in medical technology continue to unveil promising solutions, and the latest development from University of Iowa Health Care researchers is no exception. A newly designed device, about the size of a dime, promises to transform the treatment experience for bladder cancer patients, offering a less invasive and more convenient method of drug delivery.
This breakthrough device, dubbed DRIFT (Drug-Releasing Intravesical Floating Technology), is specifically engineered to administer chemotherapy drugs to patients with non-muscle-invasive bladder cancer (NMIBC). Traditionally, patients undergoing the gemcitabine and docetaxel (gem/doce) chemotherapy regimen face lengthy clinical visits and invasive procedures. These visits can stretch up to four hours, requiring patients to remain relatively immobile, which poses challenges for treatment adherence.
The DRIFT device, however, aims to simplify this process. Developed under the guidance of James Byrne, MD, PhD, an assistant professor of radiation oncology, this flexible, 3D-printed tube is designed to float freely in the bladder, delivering the docetaxel drug in a timely manner. Byrne, also affiliated with the UI Health Care Holden Comprehensive Cancer Center, explains, “Our hope is that this simple device could transform the patient experience.”
Revolutionizing Bladder Cancer Treatment
The innovative DRIFT device is inserted into the bladder through a standard foley catheter. Once inside, it releases docetaxel after the prior administration of gemcitabine has been flushed out naturally. This allows patients to continue their daily activities after a brief clinic visit. The device is then easily removed at home by the patient using an attached string.
Byrne and his team have tested this technology in animal models, demonstrating its efficacy in maintaining drug containment during the initial treatment phase. According to a recent study published in the journal BJU International (BJUI), the device’s design ensures continuous urine flow and prevents urethral obstruction. The next steps involve further pharmacokinetic and safety studies before moving on to human clinical trials.
Beyond its current application with the gem/doce regimen, Byrne and his team are optimistic about the DRIFT device’s potential in treating other bladder-related conditions, such as interstitial cystitis and recurrent urinary tract infections. The research, which received funding from the National Cancer Institute and the American Cancer Society, also involved contributions from researchers at Duke University, Chongqing Medical University in China, and Rutgers Cancer Institute.
James Byrne emphasizes the potential impact: “Creating a technology that allows patients to spend less time in the clinic, with less invasive procedures, could improve quality of life for people undergoing treatment for bladder cancer.”
Bladder cancer ranks among the most prevalent cancers in the U.S. The DRIFT device holds the promise of benefitting the 75% of patients diagnosed with early-stage non-muscle-invasive bladder cancer (NMIBC), a condition that is typically treatable yet demands time-consuming and invasive treatment methods.
Read More Here